NA Labetalol Hydrochloride Impurity - Anax Laboratories
Por um escritor misterioso
Descrição
Anax Laboratories provides Chemical industry users with Impurities of Labetalol Hydrochloride Impurity(NA) Boiling point Melting point, Labetalol Hydrochloride Impurity (NA ) Density MSDS Formula Use,If You also need to Labetalol Hydrochloride Impurity (NA ) Other information,welcome to contact us.

Amneal Pharmaceuticals LLC Issues Voluntary Nationwide Recall of Metformin Hydrochloride Extended Release Tablets, USP, 500 mg and 750 mg, Due to Detection of N-Nitrosodimethylamine (NDMA) Impurity

TODO ORO BAJA CALIFORNIA 5
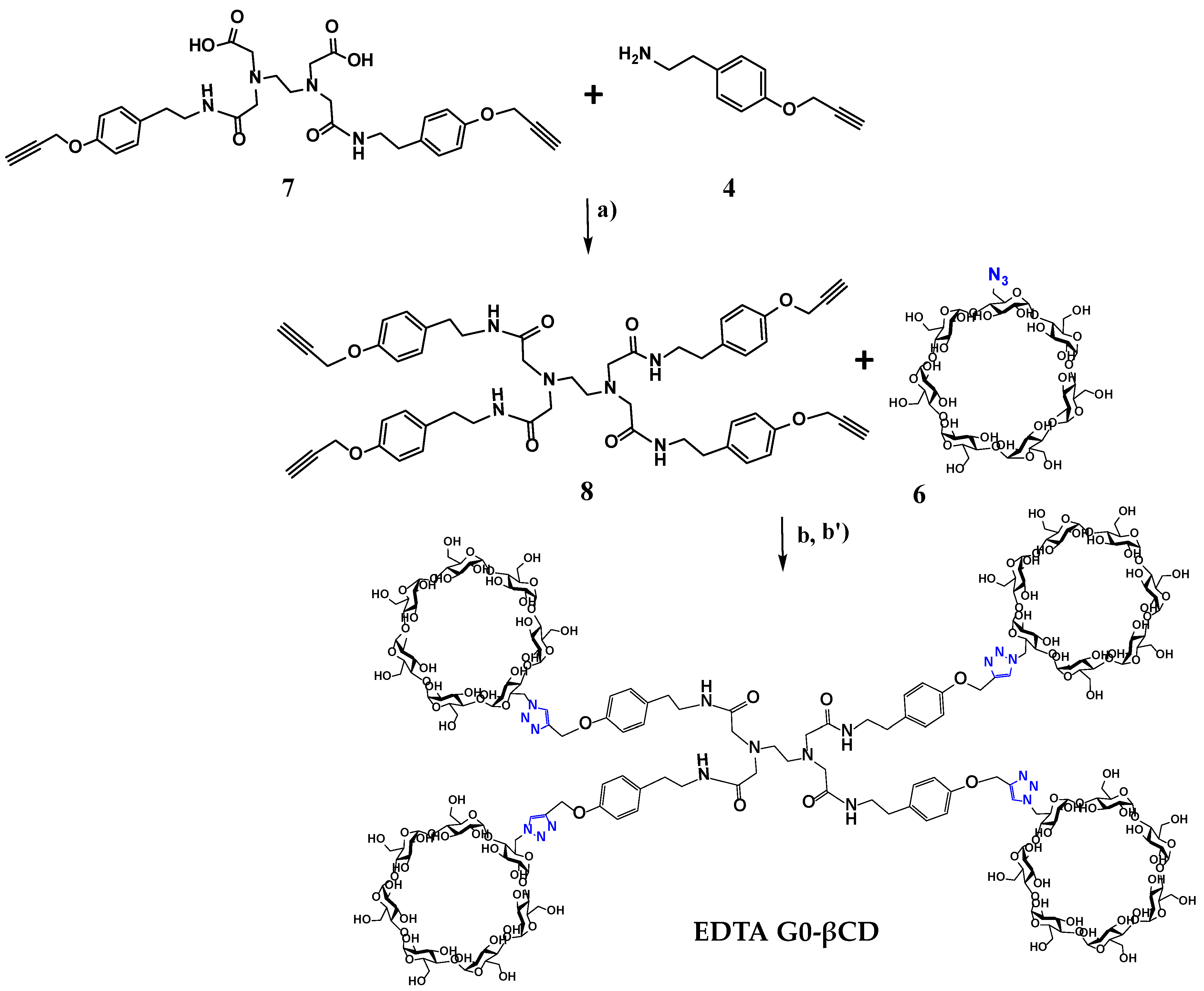
Pharmaceutics, Free Full-Text

RACK R2C-18U

Namal Jayathilake
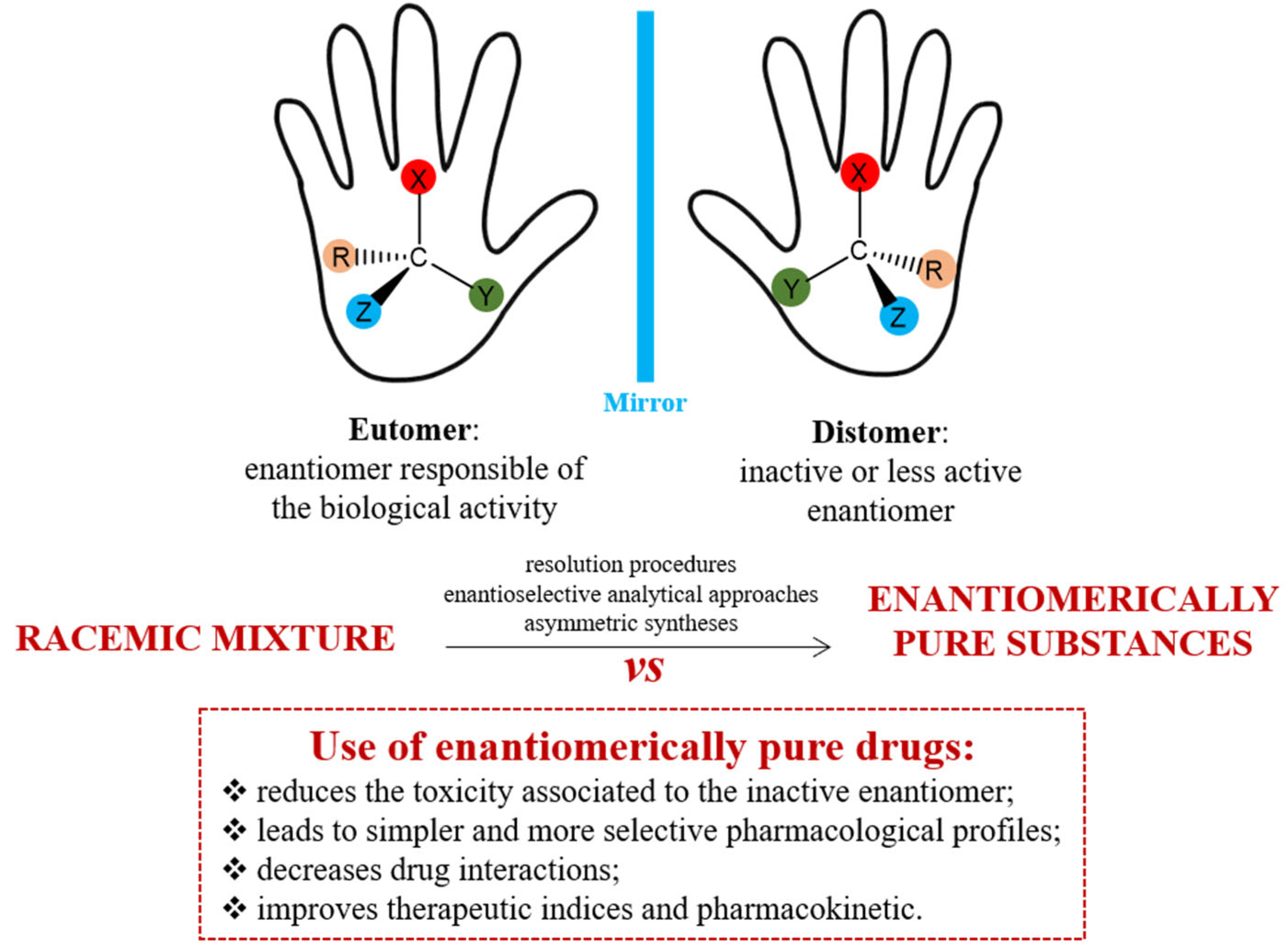
Applied Sciences, Free Full-Text
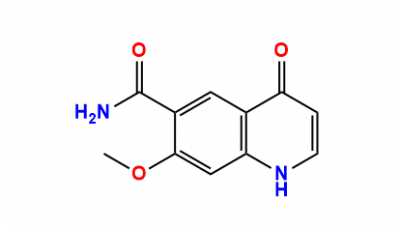
Impurities Index, CAS Number, Product Code
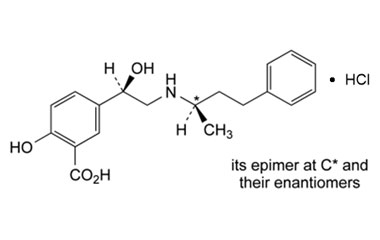
CAS NO : 2726492-68-6, Product Name : Labetalol Hydrochloride - Impurity A (Hydrochloride Salt), Chemical Name : Mixture of 4 stereoisomers of 2-hydroxy-5-[1-hydroxy-2-[(1-methyl-3-phenylpropyl)amino]ethyl]benzoic Acid Hydrochloride

126110-99-4 Fosinopril Sodium Impurity - Anax Laboratories
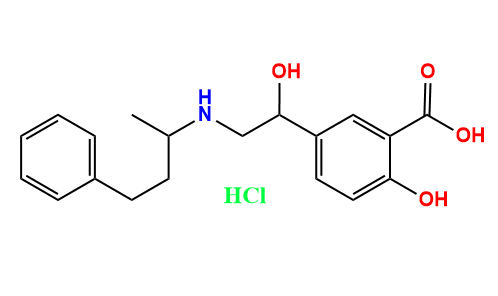
NA Labetalol Hydrochloride Impurity - Anax Laboratories
Pfizer Hospital US
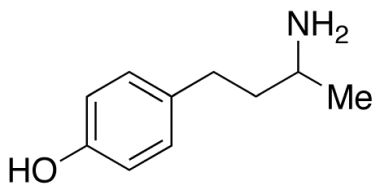
CAS NO : 2726492-68-6, Product Name : Labetalol Hydrochloride - Impurity A (Hydrochloride Salt), Chemical Name : Mixture of 4 stereoisomers of 2-hydroxy-5-[1-hydroxy-2-[(1-methyl-3-phenylpropyl)amino]ethyl]benzoic Acid Hydrochloride
de
por adulto (o preço varia de acordo com o tamanho do grupo)